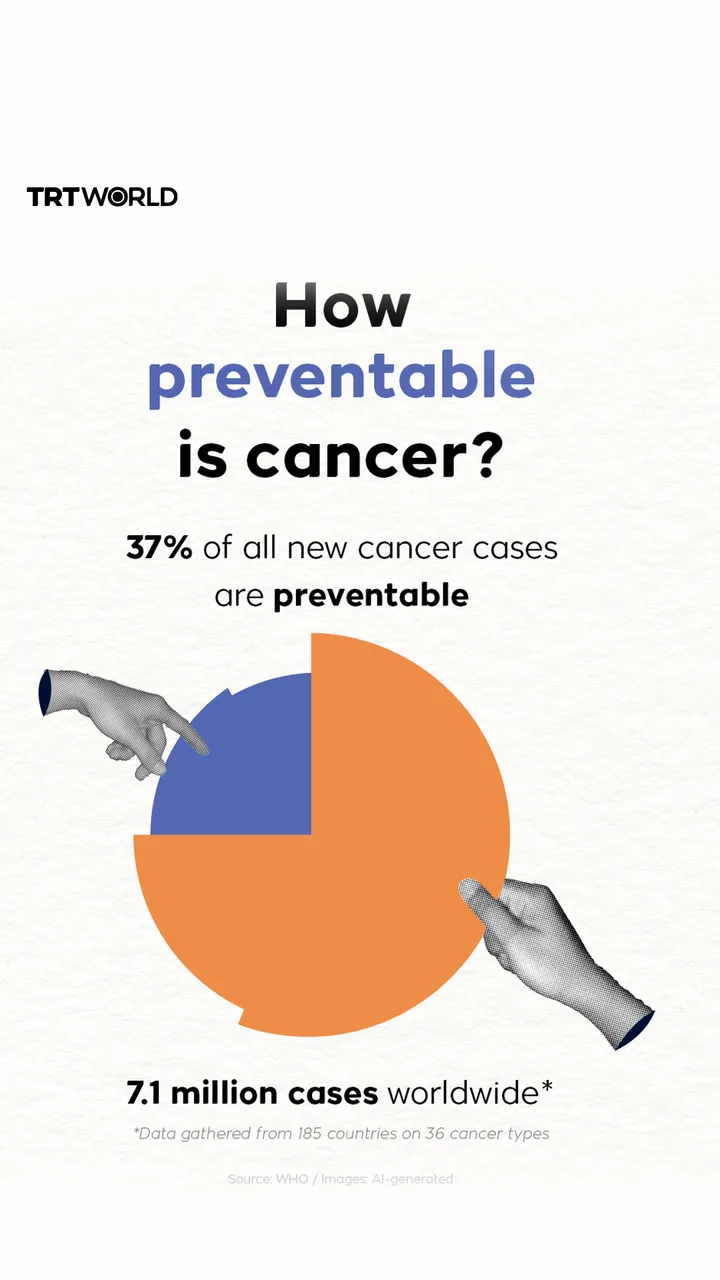
Russia's Sputnik V coronavirus vaccine is 92 percent effective according to initial test results, its developers have said. as Moscow races against its competitors in the West.
The calculations were based on results from 16,000 individuals who received both doses of the vaccine, Russia's Health Ministry, the state-run Gamaleya research centre, and the Russian Direct Investment Fund (RDIF) said in a statement on Wednesday.
"The Sputnik V vaccine had an efficacy rate of 92 percent after the second dose," the statement said, after 20 of the volunteers – some of whom were given the placebo – tested positive for coronavirus.
The adenovirus vector-based vaccine uses modified viruses of the regular flu.
Some of those vaccinated experienced "pain at the injection site, flu-like syndrome including fever, weakness, fatigue, and headache," the statement said.
First country to register vaccine
Russia in August became the first country to register a coronavirus vaccine but did so ahead of the large-scale clinical trials that are still underway.
Forty thousand volunteers at 29 medical centres are taking part in the third and final phase of Sputnik V trials.
In September, the vaccine was separately administered to medics and other at-risk people working in Russian hospitals, demonstrating an efficacy rate of "over 90 percent," the statement said.
Regional authorities in Siberia's Altai region on Tuesday said that at least three medics out of the 42 that received the vaccine had contracted the virus.
Research data to be published
Wednesday's statement added that the interim research data will be published in one of the world's "leading peer-reviewed medical academic journals".
Observation of trial participants will continue for six months, after which the full clinical trial report will be presented, it added.
The statement said that RDIF, which is funding the development of Sputnik V, will provide research data to countries that are interested in purchasing the Russian vaccine.
Overseas trials are also taking place in the UAE, Venezuela, Belarus, and other countries, the statement said.
The announcement of Sputnik V's interim results comes days after Western vaccine developers Pfizer and BioNTech said that their vaccine was more than 90 percent effective.
Geopolitical importance
The Russian drug is named Sputnik V after the Soviet-era satellite that triggered the space race, a nod to the project's geopolitical importance for Russian President Vladimir Putin.
Russia registered the vaccine for public use in August, the first country to do so, ahead of the start of the large-scale trial in September.
So far, it has inoculated 10,000 members of the public considered at high risk of contracting Covid-19 such as doctors and teachers, outside of the trial.